BOSTON, Massachusetts and HAMILTON, Ontario—February 19, 2019- Fusion Pharmaceuticals, a biopharmaceutical company focused on radiotherapeutics (specifically, targeted alpha therapeutics), today announced that the first patient was dosed in a Phase 1, first-in-human clinical trial of lead product candidate [225Ac]-FPI-1434 in patients with advanced solid tumors.
“The entire Fusion team takes tremendous pride in having developed a new targeted alpha therapeutic and in moving it from the discovery phase to a clinical trial,” said John Valliant, Ph.D., Founder and Chief Executive Officer of Fusion Pharmaceuticals. “We are excited to start clinical development of a new generation of radiopharmaceuticals utilizing our Fast-Clear™ Linker to address the need for better cancer treatments.”
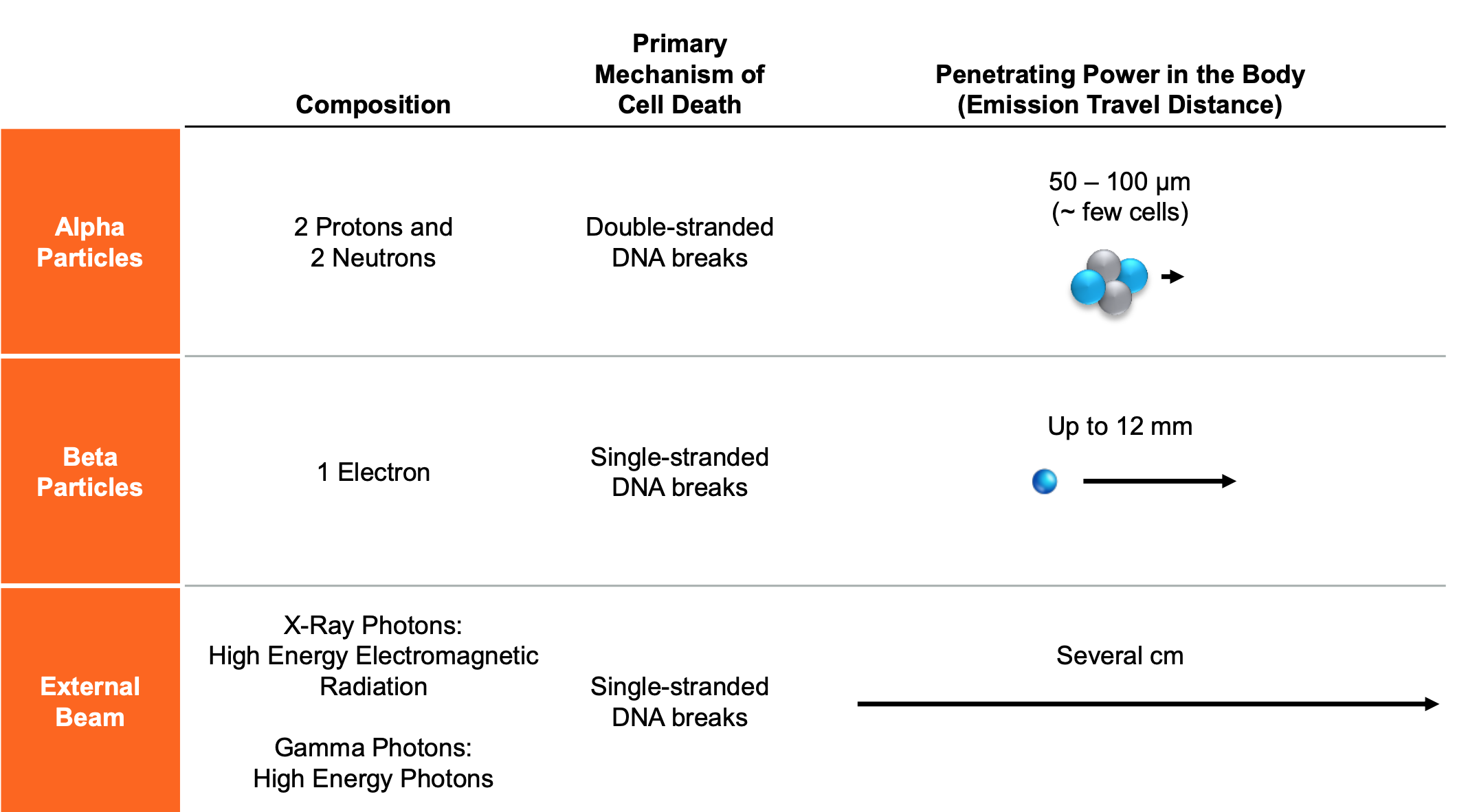
The Phase 1 clinical study (FPX-01-01) is a dose-escalation study that will characterize the safety and tolerability of a single dose [225Ac]-FPI-1434 Injection in patients with advanced solid tumors. [225Ac]-FPI-1434 is a targeted alpha radioimmunoconjugate that consists of a humanized monoclonal antibody targeting the insulin-like growth factor-1 receptor 1 (IGF-1R), Fusion’s Fast-Clear™ Linker, a bifunctional chelate, and actinium-225. Patients will be screened for target expression with the imaging agent [111In]-FPI-1547. [111In]-FPI-1547 contains the same targeting antibody, Linker, and bifunctional chelate as the [225Ac]-FPI-1434 therapeutic form, however it will contain indium-111 in place of actinium-225. Only those patients who have IGF-1R expression will be receiving the [225Ac]-FPI-1434 therapeutic radioimmunoconjugate. Investigators will evaluate the safety and tolerability of a targeted alpha-emitting radiotherapeutic. Up to 30 patients will be treated and followed for safety and for signs of efficacy. For more information about this study, visit: https://clinicaltrials.gov/ct2/show/NCT03746431
About Fusion’s Fast-Clear™ Technology Platform and FPI-1434
Fusion uses its proprietary Fast-Clear™ Technology Platform to convert molecules into radiopharmaceuticals. Fast-Clear™ linkers promote increased clearance of radoisotopes – the linkers are engineered to improve the safety of radiopharmaceuticals without impacting efficacy. The predecessor [225Ac]-FPI-1434 antibody (AVE1642) is a humanized antibody against IGF-1R (insulin-like growth factor receptor) that had previously been in Phase I and II clinical trials. Fusion has an exclusive license to AVE1642 from ImmunoGen, Inc. Fusion applied its Fast-Clear™ linker to convert AVE1642 into a potent radiopharmaceutical, [225Ac]-FPI-1434.
Acknowledgement of US DOE and 225Ac Supply
The 225Ac used in this research was supplied by the United States Department of Energy Office of Science by the Isotope Program in the Office of Nuclear Physics.
About Fusion Pharmaceuticals, Inc.
Fusion Pharmaceuticals is a biopharmaceutical company located in Hamilton, Ontario, Canada and Boston, MA, focused on becoming the leader in the targeted alpha therapy field. Fusion will exploit its unique expertise in linking medical isotopes to targeting molecules to create highly effective therapeutics. In addition to its lead program, [225Ac]-FPI-1434, Fusion is expanding its pipeline of products through a proprietary protein discovery platform, in-licensing targeting molecules, and forming new strategic partnerships. For more information, please visit www.fusionpharma.com or contact info@fusionpharma.com.
Contacts
Investors
Fusion Pharmaceuticals
Lynn Wick
info@fusionpharma.com
Media
KKH Advisors
Kimberly Ha
kimberly.ha@kkhadvisors.com
917-291-5744